|
He remained for seven years, until the college's worsening financial situation led to his resignation. At 27, he was appointed teacher of mathematics and natural philosophy at the "Manchester Academy" in Manchester, a dissenting academy (the lineal predecessor, following a number of changes of location, of Harris Manchester College, Oxford). He acquired much scientific knowledge from informal instruction by John Gough, a blind philosopher who was gifted in the sciences and arts. Around the age of 23, Dalton may have considered studying law or medicine, but his relatives did not encourage him, perhaps because being a Dissenter, he was barred from attending English universities. When he was 15, Dalton joined his older brother Jonathan in running a Quaker school in Kendal, Westmorland, about 45 miles (72 km) from his home. ( May 2021) ( Learn how and when to remove this template message) Unsourced material may be challenged and removed. 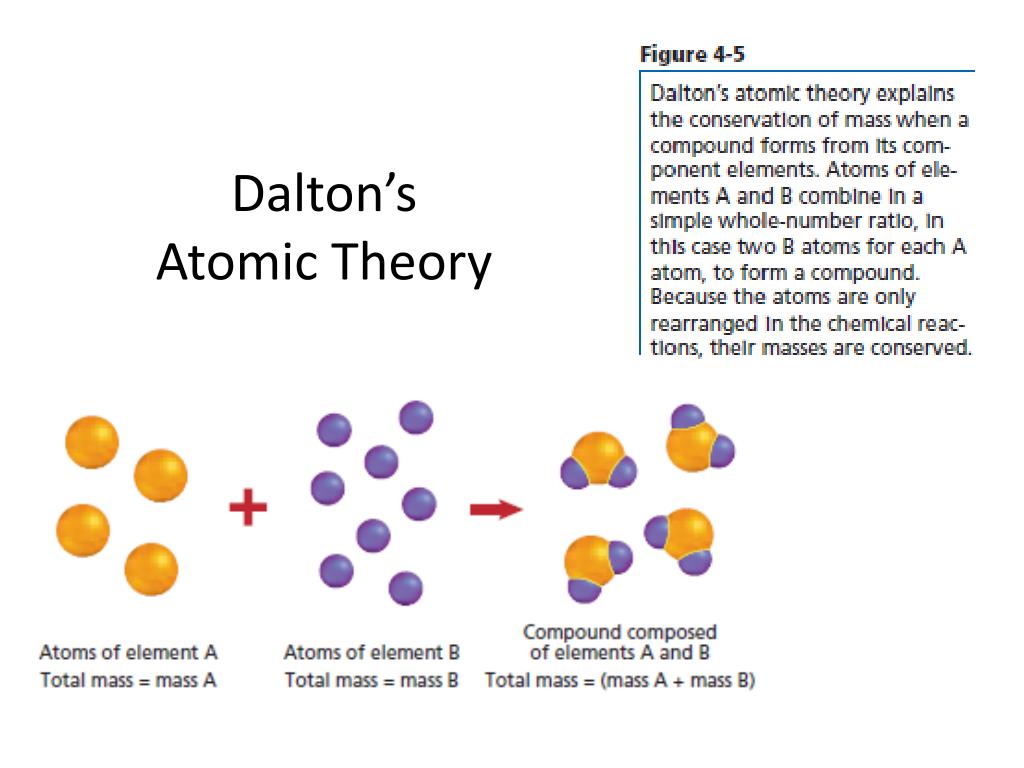
Please help improve this section by adding citations to reliable sources. Dalton's family was too poor to support him for long and he began to earn his living, from the age of ten, in the service of wealthy local Quaker Elihu Robinson. He received his early education from his father and from Quaker John Fletcher, who ran a private school in the nearby village of Pardshaw Hall. John Dalton was born into a Quaker family in Eaglesfield, near Cockermouth, in Cumberland, England. Early life Historical plaque marking birthplace of John Dalton Modern plaque marking birthplace of John Dalton He also researched colour blindness, which he had as a result, colour blindness is known as Daltonism in several languages. 
He introduced the atomic theory into chemistry. Print.John Dalton FRS ( / ˈ d ɔː l t ən/ 5 or 6 September 1766 – 27 July 1844) was an English chemist, physicist and meteorologist. 
Upper Saddle River, NJ: Pearson/Prentice Hall, 2008. Again, this is true in chemical reactions but not necessarily in a nuclear reaction. In a chemical reaction, atoms only change the way that they are bound together with other atoms. Atoms of one element cannot change into atoms of another element. Atoms combine in simple, whole-number ratios to form compounds.This directly relates to Proust's Law of Definite Proportions.Ĥ. "All atomes of a given element have the same atomic number" would be a more correct statement.ģ. All atoms of a given element have the same mass and other properties that distinguish them from the atoms of other elements. This theory was later proved to be partially incorrect isotopes are atoms of the same element that differ in weight due to varying numbers of neutrons in their nucleus. Exploration in nuclear chemistry has revealed that it is possible to destroy atoms however, atoms remain indestructible during chemical reactions.ĭalton's Atomic Theory vs. Today, we know that the even atoms are made up of smaller particles called protons, neutrons, and electrons. Each element is composed of tiny, indestructible particles called atoms. This idea dated back to the Greeks and Democritus's theories about matter. Based on a combination of their prior knowledge and his own experiments, Dalton put together the first version of his atomic theory which stated the following ideas.ġ. 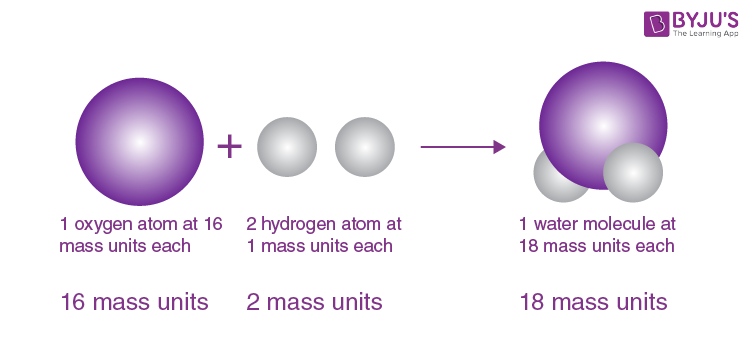
Dalton was mistrustful of other scientists' work and liked to assume as little as possible, but he could not have formed his atomic theory without the discoveries of Lavoisier, Priestley, and Proust.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
